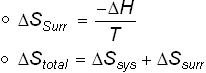
The formula you wrote is for an ideal gas, which would not normally be used as a reservoir. (According to this formula: dS=Cvln(T2/T1)+Rln(V2/V1) )īut when we solve many matters in thermodynamics especially in entropy part, we think that the reservoir exchanging the heat with another part has the entropy change expressed as Q/T not zero. In short, the Boltzmann formula shows the relationship between entropy and the number of ways the atoms or molecules of a certain kind of thermodynamic system can be arranged.

If the system loses an amount of heat q at a temperature T 1, which is received by surroundings at a temperature T 2. (1) where is the Boltzmann constant (also written as simply ) and equal to 1.380649 × 10 23 J/K, and is the natural logarithm function. Entropy change 353.8 - 596 -242.2 J K -1 mol -1 Notice that it is a negative value. Total entropy change is equal to the sum of entropy change of system and surroundings. The argument is that because the surroundings may be approximated as either constant volume or constant pressure, the heat absorbed/released by it is equal to the internal energy or enthalpy. Total entropy at the end 214 + 2 (69.9) 353.8 J K -1 mol -1 Entropy change what you end up with - what you started with. The entropy change of the surroundings can be calculated by the equation d S s u r d q T s u r regardless of the path (irreversible or reversible). Thus, the change in entropy accompanying a chemical reaction can be estimated qualitatively by considering the disorderliness of the structures of the species. Reservoir has no change in temperature and volume. Total starting entropy 186 + 2 (205) 596 J K -1 mol -1 You ended up with 1 mole of carbon dioxide and two moles of liquid water. The entropy (S) of a given system is the number of possible arrangements of the particles and their energy in a given system. The process is defined as the quantity of heat emitted or absorbed isothermally and reversibly divided by the absolute temperature during entropy change.
